PC3I News
In a new study published in JAMA Oncology, my colleagues and I find that behavioral nudges can promote high-value, evidence-based prescribing of specialty drugs in cancer care. For patients with breast, lung, and prostate cancer with bone metastases, clinicians face a decision between two therapies of comparable effectiveness but dramatically different cost. Guidelines endorse the use of either zoledronate or denosumab, bone-modifying agents that protect against skeletal complications. But zoledronate costs as little as $215 a year while denosumab costs as much as $26,000 a year.

To conduct our study, we took advantage of concurrent quality improvement nudges at 2 of 7 practice sites within University of Pennsylvania Health System. At Site A, the main cancer center, clinical leadership endorsed zoledronate (over denosumab) and presented performance feedback at quarterly meetings and via email. Site B, a community affiliate, implemented these same nudges and additionally accountable justification, in which clinicians had to justify denosumab prescription to the pharmacy. Using a quasi-experimental design, we examined the effects of these increasingly potent behavioral nudges on zoledronate prescription. Compared to five other usual care sites serving collectively as control, the two sites implementing nudges achieved dramatically higher rates of zoledronate prescription: Site A saw a 26 percentage point increase, and site B a 44.9 percentage point increase, in predicted rates of zoledronate prescription. The Figure below shows unadjusted zoledronate (vs denosumab) prescription rates over the study period.
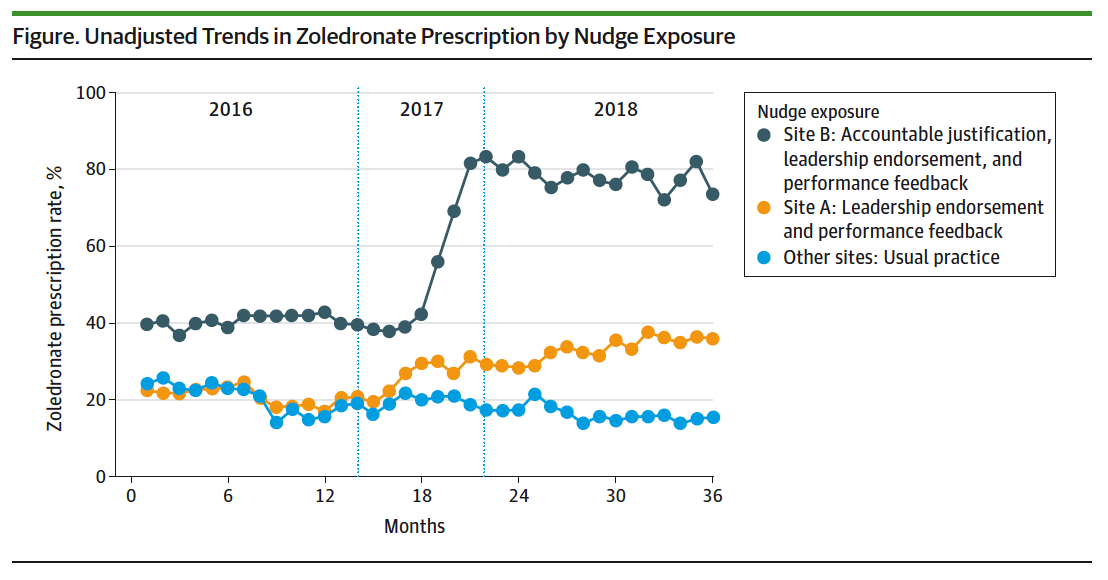
National guidelines identify hundreds of evidence-based cancer therapies, which often vary considerably in cost but yield similar outcomes. As a result, clinicians and patients frequently confront treatment decisions where one alternative affords significantly higher value than another. The stakes are particularly high in cancer care, where spending will exceed $150 billion in 2020, driven in large part by cancer drug prescribing. This study suggests an effective strategy for nudging clinicians towards higher-value cancer care.
Samuel Takvorian is an instructor in the Division of Hematology and Oncology at Penn’s Perelman School of Medicine, and a PC3I faculty member. The study, “Association of Behavioral Nudges With High-Value Evidence-Based Prescribing in Oncology” was authored by Samuel U. Takvorian, MD, MS; Vrushabh P. Ladage, MHCI; E. Paul Wileyto, PhD; Drew S. Mace, PharmD; Rinad S. Beidas, PhD; Lawrence N. Shulman, MD; and Justin E. Bekelman, MD and appeared in JAMA Oncology on April 30, 2020.
Related News
PC3I News
Cancer Service Line & PC3I Select 2024 Innovation Accelerator Project
04.22.2024
The Cancer Service Line (CSL) and the Penn Center for Cancer Care Innovation (PC3I) have announced the selection of the 2024 Penn Cancer Innovation Accelerator project. The chosen project, submitted by Katie Boyk, BS, MHA, will identify solutions to improve cancer care scheduling workflows for patients, clinicians, and staff.
PC3I News
PC3I and Tennessee Oncology’s McKay Institute for Oncology Transformation Unveil New Collaborative Relationship
02.06.2024
PC3I and Tennessee Oncology’s McKay Institute for Oncology Transformation announce an academic-community oncology innovation partnership to improve cancer care delivery.
PC3I News
PC3I Leadership Presents at 2023 AORTIC International Conference on Cancer in Africa
12.05.2023
PC3I Associate Director Surbhi Grover and Deputy Director Katharine Rendle presented research at the 14th AORTIC International Conference on Cancer in Africa, which took place in Dakar, Senegal.


